Trying to make the CPA system prototype without a finalised CPA master list is very difficult. It’s hard to work out if the results are weird because the underlying system has holes in it or because the implementation has problems. Therefore, and because it’s a task we need to do at some point anyway, I think we should make a full and final list of compounds, processes and organelles for the microbe stage. There are previous attempts at this here and here.
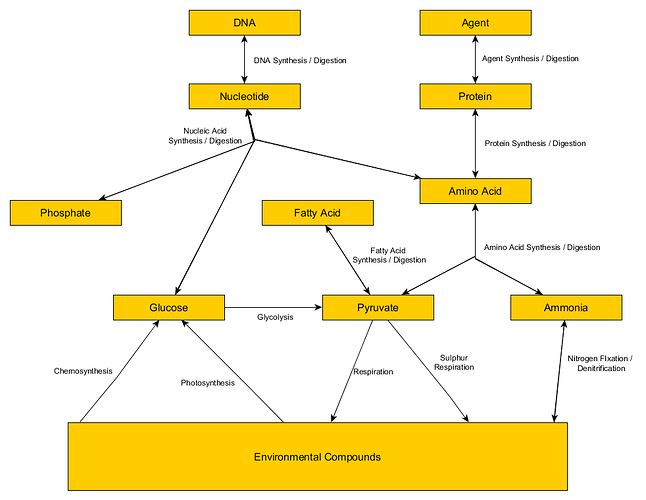
Compounds:
Environmental
Sunlight
Sulfur : S
Hydrogen Sulfide : H2S
Oxygen : O2
Nitrogen : N2
Carbon Dioxide : CO2
Ammonia : NH3
Phosphates : PO4
Manufactured by microbes.
ATP : Not specified or conserved. It’s just a currency.
Light - Bioluminescent
Glucose
Pyruvate
Amino Acids
Fat
Protein
Agents
Nucleotide
DNA
Processes:
Chemosynthesis: 6CO2 + 12 Hydrogen Sulfide -> 1 Glucose + 12 Sulfur
Photosynthesis: 6CO2 + Sunlight -> Glucose + 6O2
Glycolysis: 1 Glucose -> 2 Pyruvate + 2 ATP
Respiration: 1 Pyruvate + 3 Oxygen -> 3 CO2 + 18 ATP
Sulfur Respiration: 1 Pyruvate + 3 Sulfur -> 3 CO2 + 3 H2S + 8ATP
Protein Synthesis: 1 Amino Acid + 4 ATP -> 1 protein
Protein Digestion: 1 Protein -> 1 Amino Acid
Amino Acid Synthesis: 1 Pyruvate + 3 ATP + 1 Ammonia -> 1 Amino Acid
Amino Acid Digestion: 1 Amino Acid -> 2 ATP + 1 Pyruvate + 1 Ammonia
Fatty Acid Synthesis: 9 Pyruvate + 56 ATP -> 1 Fatty Acid + 9 CO2
Fatty Acid Digestion: 1 Fatty Acid -> 6 Pyruvate + 45 ATP
Nucleotide Synthesis: 1 Glucose + 1 Phosphate + 8 ATP + 2 Amino Acid -> 1 Nucleic Acid
Nucleotide Digestion: 1 Nucleic Acid -> 1 Glucose + 2 Amino Acid + 1 Phosphate
Agent Synthesis: 1 Protein + 5 ATP -> 1 Agent
Agent Disgestion: 1 Agent -> 1 Amino Acid
DNA Synthesis: 1 Nucleotide + 5 ATP -> 1 DNA
DNA Digestion: 1 DNA -> 1 Nucelotide
Nitrogen Fixation: 1 N2 + 16 ATP -> 2 NH3
Denitrification: 2 NH3 -> N2 + 10 ATP
Here is a chart of the above.
CPA Organelles:
Chemoplast : Chemosynthesis
Chloroplast : Photosynthesis
Cytoplasm : Glycolysis, Fat Synthesis and Digestion, Amino Acid Synthesis and Digestion
Mitochondria : Respiration
Sulphur-Mitochondria? : Sulphur Respiration
Agent Gland : Agent synthesis
Nucleus/Endoplasmic Reticulum/Golgi Body: Nucleic Acid Synthesis, DNA Synthesis, Protein Synthesis
Lysosomes: Protein Digestion, Nucleic Acid Digestion, DNA Digestion, Agent Digestion
(Therefore the minimal viable cell is Nucles/ER/GB + Cytoplasm)
Utility Organelles:
Flagella
Cilia
Pilus? (Either a hair like appendage or one of these )
Directional Agent Secretor?
Water Pump
Microbial Eye?
Engulfing Edge
Bioluminescent Gland?
Plant Cell Wall
Cytoskeleton?
Vesicle/Vacuole
(Looking through wikipedia’s list of organelles looks like we’ve got a pretty complete list)
 In the end we’ll get down to 1.
In the end we’ll get down to 1.